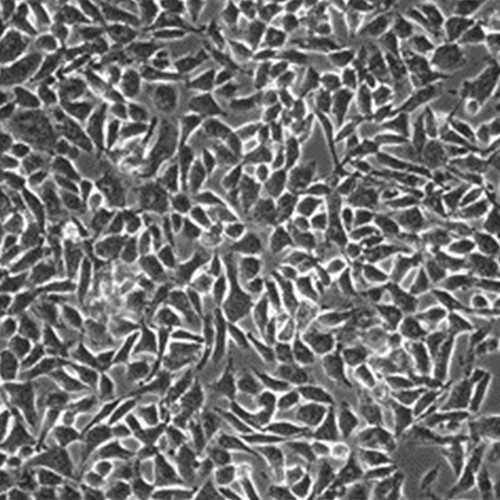
BHK-21/WI-2 cells for Generating VSV
These BHK-21 cells are highly transfectable and are used to recover infectious recombinant vesicular stomatitis virus (rVSV) as originally described by Lawson et al (5) and modified by Jayakar et al (2), or for the recovery and amplification of replication-restricted ΔG-VSV as described by Whitt (7).
The cells have been characterized by isoenzyme analysis which confirmed that the cells are from Syrian golden hamsters. The cells also have been shown to be mycoplasma-free using the Strategene Mycoplasma Plus PCR Primer Set.
From the laboratory of Michael A. Whitt, Ph.D., University of Tennessee.
 Part of The Investigator's Annexe program.
Part of The Investigator's Annexe program.
These BHK-21 cells are highly transfectable and are used to recover infectious recombinant vesicular stomatitis virus (rVSV) as originally described by Lawson et al (5) and modified by Jayakar et al (2), or for the recovery and amplification of replication-restricted ΔG-VSV as described by Whitt (7).
The cells have been characterized by isoenzyme analysis which confirmed that the cells are from Syrian golden hamsters. The cells also have been shown to be mycoplasma-free using the Strategene Mycoplasma Plus PCR Primer Set.
From the laboratory of Michael A. Whitt, Ph.D., University of Tennessee.
 Part of The Investigator's Annexe program.
Part of The Investigator's Annexe program.
Specifications
| Product Type: | Cell Line |
| Name: | BHK-21 |
| Cell Type: | Baby Hamster Kidney |
| Organism: | Mesocricetus auratus(hamster, Syrian golden) |
| Source: | Kidney |
| Morphology: | Epithelial/Fibroblast |
| Biosafety Level: | Bsl-1 |
| Growth Conditions: | DMEM + 5% FBS; 37oC, 6-8% CO2 |
| Subculturing: | Passage 1:5-1:3 every day, or 1:20 every 2-3 days. |
| Cryopreservation: | 70% DMEM; 20% FBS; 10% DMSO |
| Comments: | For suggested protocol, see: Whitt, MA, J. Virol. Methods, 2010. 169(2): p. 365-74. |
| Storage: | Liquid nitrogen |
| Shipped: | Dry ice |
Provider
From the laboratory of Michael A. Whitt, Ph.D., University of Tennessee.
References
- Helenius, A., and H. Soderlund. 1973. Stepwise dissociation of the Semliki Forest virus membrane with Triton X-100. Biochem, Biophys.Acta 307:287-300.
- Jayakar, H. R., K. G. Murti, and M. A. Whitt. 2000. Mutations in the PPPY motif of vesicular stomatitis virus matrix protein reduce virus budding by inhibiting a late step in virion release. J. Virol 74:9818-9827.
- Vaheri A, Sedwick WD, Plotkin SA, Maes R. Cytopathic effect of rubella virus in BHK21 cells and growth to high titers in suspension culture. Virology. 1965 Oct;27(2):239-41.
- Kaarianen, L., K. Simons, and C.-H. von Bonsdorff. 1969. Studies in subviral components of Semliki Forest virus. Ann.Med.Exp.Fenn 47:235-248.
- Lawson, N. D., E. A. Stillman, M. A. Whitt, and J. K. Rose. 1995. Recombinant vesicular stomatitis viruses from DNA. Proc.Natl.Acad.Sci.(USA) 92:4477-4481.
- Macpherson, I., and M. Stoker. 1962. Polyoma transformation of hamster cell clones - an investigation of genetic factors affecting cell competence. Virology 16:147-151.
- Whitt, M. A. 2010. Generation of VSV pseudotypes using recombinant DeltaG-VSV for studies on virus entry, identification of entry inhibitors, and immune responses to vaccines. J. Virol. Methods 169:365-74.
- Suarez MA, Valencia J, Cadena CC, Maiti R, Datta C, Puerto G, Isaza JH, San Juan H, Nagaraja V, Guzman JD. Diarylethenes Display In Vitro Anti-TB Activity and Are Efficient Hits Targeting the Mycobacterium tuberculosis HU Protein. Molecules. 2017 Jul 25;22(8). pii: E1245. View Article
- Zeng Z, Yu H, Chen H, Qi W, Chen L, Chen G, Yan W, Chen T, Ning Q, Han M, Wu D. Longitudinal changes of inflammatory parameters and their correlation with disease severity and outcomes in patients with COVID-19 from Wuhan, China. Crit Care. 2020 Aug 27;24(1):525. View article
- Galindo I, Garaigorta U, Lasala F, Cuesta-Geijo MA, Bueno P, Gil C, Delgado R, Gastaminza P, Alonso C. Antiviral drugs targeting endosomal membrane proteins inhibit distant animal and human pathogenic viruses. Antiviral Res. 2020 Nov 26:104990. View article
- Hashem AM, Algaissi A, Almahboub SA, Alfaleh MA, Abujamel TS, Alamri SS, Alluhaybi KA, Hobani HI, AlHarbi RH, Alsulaiman RM, ElAssouli MZ, Hala S, Alharbi NK, Alhabbab RY, AlSaieedi AA, Abdulaal WH, Bukhari A, Al-Somali AA, Alofi FS, Khogeer AA, Pain A, Alkayyal AA, Almontashiri NAM, Ahmad BM, Li X. Early Humoral Response Correlates with Disease Severity and Outcomes in COVID-19 Patients. Viruses. 2020 Dec 4;12(12):E1390. View article
- Almahboub SA, Algaissi A, Alfaleh MA, ElAssouli MZ, Hashem AM. Evaluation of Neutralizing Antibodies Against Highly Pathogenic Coronaviruses: A Detailed Protocol for a Rapid Evaluation of Neutralizing Antibodies Using Vesicular Stomatitis Virus Pseudovirus-Based Assay. Front Microbiol. 2020 Sep 4;11:2020. View Article
- Farzani TA, Chov A, Herschhorn A. A protocol for displaying viral envelope glycoproteins on the surface of vesicular stomatitis viruses. STAR Protoc. 2020 Dec 9;1(3):100209. View article
- Wu K, Werner AP, Moliva JI, Koch M, Choi A, Stewart-Jones GBE, Bennett H, Boyoglu-Barnum S, Shi W, Graham BS, Carfi A, Corbett KS, Seder RA, Edwards DK. mRNA-1273 vaccine induces neutralizing antibodies against spike mutants from global SARS-CoV-2 variants. bioRxiv [Preprint]. 2021 Jan 25:2021.01.25.427948. View article
- Baggen J, Persoons L, Vanstreels E, Jansen S, Van Looveren D, Boeckx B, Geudens V, De Man J, Jochmans D, Wauters J, Wauters E, Vanaudenaerde BM, Lambrechts D, Neyts J, Dallmeier K, Thibaut HJ, Jacquemyn M, Maes P, Daelemans D. Genome-wide CRISPR screening identifies TMEM106B as a proviral host factor for SARS-CoV-2. Nat Genet. 2021 Apr;53(4):435-444. View article
If you publish research with this product, please let us know so we can cite your paper.